Surfactants have unique structural features that make them useful in many industrial applications, many of them have found their places in our major household uses. Surfactants and its behavior in solutions is a fast-paced developing field and has application in a variety of industries, medical and life sciences, analytical chemistry, physics, and engineering. All these industries and fields, in one way or another, contribute to pollution. [1][2][3]
Amphiphilic molecules have hydrophilic and lipophilic units that improve interfacial properties. Surfactant at low concentrations in the systems adsorbs onto the surfaces or interfaces alter the surface or interfacial free energy of those surfaces/ interfaces. ‘Interface’ is a boundary between two immiscible phases whereas ‘Surface’ stands for an interface where one phase is a gas, generally air. This characteristic of surfactant makes it suitable for the fields of detergent, wetting, emulsification, oil recovery, froth flotation, and other fields. (Muntaha and Khan, 2015)
A huge number of synthetic surfactants used in industrial and domestic work are diffused in different environmental parts, such as water, soil, sediment, etc. Studies have shown that more than 3/5th of surfactant derivatives are released into the aquatic environment. (Naylor et al., 1992; Schmitt et al.,2014). Global surfactant production was approximately 12.5 billion kg in 2006 (Edser, 2006), while Western Europe produced more than 3 billion kg in 2007. (Ivankovic and hrenovic, 2010). In 2010, the use of non-ionic surfactants (polyethoxylated nonylphenol) in the United States was approximately 172,000 tons. (Schmitt et al., 2014) These non-biodegradable surfactants can disturb the climate and cause health risks such as respiratory tract, dermatitis, eye irritation, etc. (Tmakova et al., 2015; Pradhan and Bhattacharya, 2014).
Providing more evidence to the negative effect of Non-biodegradable surfactants, Ostroumov expressed how non-biodegradable surfactants and their derivatives hinder the filtration activities of oysters and mussels. Cationic surfactants have the most toxic effect on microbes followed by anionic and non-ionic surfactants. (Singh et al., 2002). Non- biodegradable surfactants have a similarly damaging effect on microorganisms, aquatic plants, humans, and other elements of nature. (Cserhati et al.,2002).
An increase in large scale production of surfactants due to technological advancements have also serious environmental concerns. (Chevalier, 2002). With these concerning environmental effects, it is desirable that surfactants are biodegradable and less toxic while being surface active.[4][5][6] Thus, the quest for environment-friendly surfactants has begun. These surfactants could be either natural surfactants or synthetic biodegradable surfactants. Different techniques like extraction, distillation, filtration, etc are required to get natural surfactants that are directly obtained from plants, trees, bacteria, fungi, and other natural sources. As an alternative to synthetic surfactants, Holmberg has reported amides of amino acids as natural surfactants and Biomass extracted Humic acid has been reported as a surfactant by Salati et al.
A recent trend in industries is to avoid synthetic surfactants. With this view, we have studied tea Saponion with an objective to evaluate its micellar and interfacial properties like CMC, surface tension, foaming, cleaning, and pH. As a reference, we have also studied commercial synthetic surfactant, which is ionic in nature. Natural Surfactant has been found equally effective that of synthetic surfactant due to the presence of the Saponion molecule in it. Molgaard et al. have observed complete biodegradation of a certain Saponion molecule in 10 days. Rahman and Gapke note that these Saponion can be economically produced at large scale and can be used in industrial environmental control activities.
Raw saponin product derived from tea was studied as a primary examination. Surfactant molecules in absolute purity are difficult to get and is a costly process. The majority of surfactant studies are in one or other form mixed surfactant systems with one or two having a higher share. Based on data obtained from comparison, other additives are introduced to get the required properties.
Surface Tension
The effectiveness of surfactant (πCMC) is given by πCMC where g0 is the surface tension of pure water and πCMC is the surface tension of solution at CMC. Natural surfactant shows maximum effectiveness followed by Synthetic.
πCMC = g0 - gCMC
Name of Surfactant | CMC (wt %) | gCMC | πCMC |
Surf excel (Surf) | 0.07 | 31.5 | 40.5 |
Tea saponin (Tea) | 0.04 | 31.4 | 40.6 |
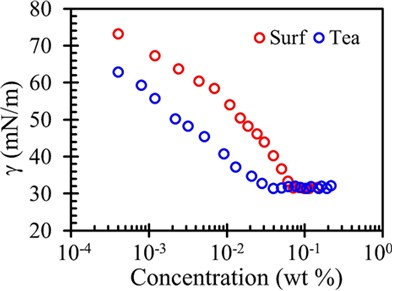
Fig 1
Foaming Behaviour
Foaming behavior and cleaning action is not much interrelated, but foam behavior is a significant aspect of cleansing agent assessment by the consumers. Foam creation and durability are imperative in numerous applications. Foam ability is the amount of foam creation due to the constant formation of new interfaces. The higher power of foaming requires faster adsorption, high surface elasticity.
Fig 2
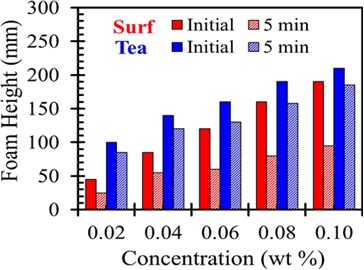
The tea solution produces dense, high- quality foam, which may be due to the presence of large amounts of saponin molecule. The presence of saponin results in a highly dynamic reduction in surface tension and helps to create the large surface required for foaming.
We have also observed under the microscope to get insight into the bubble size and foam structures as depicted in Fig.3. The foam generated from Surf contained a higher liquid portion and large bubble sizes. While Tea produces foam with low liquid portion and much smaller bubble sizes. This observation indicates that natural surfactants could serve as a good foaming agent for several applications.
Figure 3

pH and Conductivity
The investigation of pH is an essential characteristic of surfactant research (Tmakova et al., 2015). The pH of a solution changes the net charge in the head group, which leads to a change in the repulsive force between them.
There are some surfactants that have a charge, due to their charge it shows the conductance. The conductance depends on the surfactants, whether the surfactant is anionic, cationic, or zwitterionic. The change in conductivity at lower concentrations is almost constant further increases at higher concentrations for mostly surfactant. At low level of surfactant concentration, the headgroup of the surfactant is enclosed by water media, resulting in low conductivity. In addition, conductivity starts increases with the concentration of surfactant molecules due to ionization. Conductivity also correlates with mildness these are inversely proportional to each other.
These pH values for both surfactants in a concentration range of 0.02 to 0.10% by weight of the solutions prepared in Milli Q water with a pH of 6.7 ± 0.2 were investigated. The pH values of the surfactant solution at various concentrations are shown in table below. Tea with an acidic pH, possibly due to the hydrolysis of nonionic glucuronic groups, can be made of appropriate pH with suitable additives. This pH of the solution near the skin (~ 5.5) causes less damage to the hair and skin. The surf shows alkaline in nature. The pH of the tea solution decreases with concentration, while that of Surf increases.
Table The values of pH at different concentrations of surfactants solutions.
Concentration (wt %) | Surf | Tea |
0.02 | 10.2 | 5.3 |
0.04 | 10.3 | 5.2 |
0.06 | 10.5 | 5.5 |
0.08 | 10.8 | 5.9 |
0.10 | 11.2 | 6.1 |
Cleaning
Cleansing activity means, the removal of unwanted substances such as soil, grease and dirt, is the main target of any detergent. The cleaning activity was calculated by the below equation
Figure 4
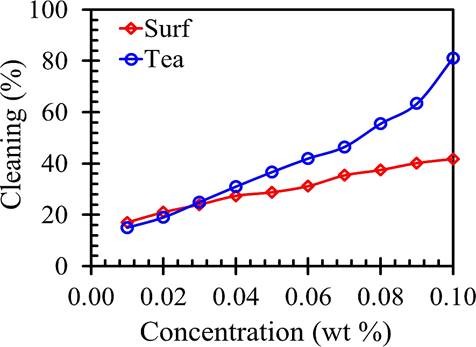
Herbal saponins, tea, were studied to find alternatives to synthetic surfactants that are commonly used and were make comparisons with marketed available surfactants (Surf Excel). The outcomes demonstrated that saponins are naturally acidic and decomposable. Natural materials are considered to be biodegradable as plant extracts. The tea saponin, which was probably examined by the first time, shows a good effectiveness, in addition to high foaming capacity, decent cleaning capacity and ultra-soft. Although lots of works account for isolation, characterization, etc. Tea Saponins are good emulsifiers and can find some industrial uses. We also quantified the cleaning capacity of surfactant solutions. Therefore, our studies can offer a simple and inexpensive method to measure the general cleaning method for the evaluation of detergents. We conclude that tea saponin comes with good surface-active properties. These studies can offer useful information for the cosmetics industry as plant-based material is a biodegradable organic surfactant. We propose a new biodegradable alternative from a plant-based material that acts as a surfactant and its systems.